Bortezomib For Injection IP
Bortezomib For Injection IP Specification
- Origin
- India
- Indication
- Multiple Myeloma and Mantle Cell Lymphoma
- Salt Composition
- Bortezomib
- Dosage Form
- Tablet
- Enzyme Types
- Other
- Feature
- Sterile, Single-Use Vial
- Ingredients
- Each vial contains 2 mg Bortezomib
- Application
- For Intravenous or Subcutaneous Use
- Physical Color/Texture
- White to Off-White Lyophilized Powder
- Fermentation Smell
- Odorless
- Enzymatic Activity
- Inhibits 26S Proteasome Activity
- Storage Instructions
- Cool & Dry Place
- Shelf Life
- 24 months
- Product Code
- Refer to packaging for exact code
- Packaging Type
- Vial with Flip-Off Seal
- Administration
- Prepared as Solution and Administered via IV or SC
- Reconstitution Solution
- 0.9% Sodium Chloride Injection
- Contraindications
- Hypersensitivity to Bortezomib or Boron
- Prescription Status
- Prescription Only Medication
- Strength
- 2 mg/vial
- Route of Administration
- Intravenous (IV) or Subcutaneous (SC)
Bortezomib For Injection IP Trade Information
- Minimum Order Quantity
- 500 Pieces
- Supply Ability
- 100 Pieces Per Day
- Delivery Time
- 3-4 Days
About Bortezomib For Injection IP
By keeping track with the current market growth, we are offering Bortezomib For Injection IP. This medication is used to treat people with multiple myeloma. Moreover, Bortezomib For Injection IP comes in a class of medications called antineoplastic agents. Apart from this, the offered medication is tested on different measures before delivering at customers end.
Effective Treatment for Serious Hematological Cancers
Bortezomib For Injection IP is a crucial therapeutic option for patients with Multiple Myeloma and Mantle Cell Lymphoma. By inhibiting 26S proteasome activity, it helps in reducing cancer cell proliferation, thus enhancing treatment outcomes. Prepared as a sterile solution, it offers flexibility in administration through intravenous or subcutaneous routes, ensuring adaptability to patient needs.
Sterile, Convenient, and Reliable Formulation
Each vial is produced under stringent quality standards, ensuring sterility and consistency. Its easy-to-reconstitute lyophilized form and flip-off seal design facilitate quick and safe preparation. Physicians and pharmacists benefit from clear packaging and instructions, promoting accurate dosing and minimizing the risk of contamination during handling.
FAQ's of Bortezomib For Injection IP:
Q: How should Bortezomib For Injection IP be prepared for administration?
A: Reconstitute the lyophilized product using 0.9% Sodium Chloride Injection as per the instructions provided. Once dissolved, the solution is ready for administration either intravenously or subcutaneously by a healthcare professional.Q: What are the main indications for using this medication?
A: Bortezomib For Injection IP is primarily used in the treatment of Multiple Myeloma and Mantle Cell Lymphoma. It works by inhibiting 26S proteasome activity, reducing the growth of malignant cells.Q: When is Bortezomib contraindicated?
A: This medication should not be used in patients who have a known hypersensitivity to Bortezomib, boron, or any of the excipients in the formulation. Consult your physician before use if you have a history of allergic reactions to similar agents.Q: Where should Bortezomib For Injection IP be stored?
A: Store the vials in a cool, dry place as per the storage instructions. Avoid exposure to direct light or excessive heat to preserve the efficacy and stability of the medication.Q: What is the process for administering Bortezomib For Injection IP?
A: After reconstitution, the solution should be administered by a qualified healthcare provider either through intravenous (IV) infusion or subcutaneous (SC) injection, depending on the patient's needs and doctor's recommendation.Q: How does this medication benefit patients with cancer?
A: By inhibiting the 26S proteasome, Bortezomib interferes with pathways critical for cancer cell survival, helping to control disease progression and improve quality of life for patients with Multiple Myeloma and Mantle Cell Lymphoma.Q: Is Bortezomib For Injection IP available over the counter?
A: No, Bortezomib For Injection IP is a prescription-only medication and must be administered under medical supervision.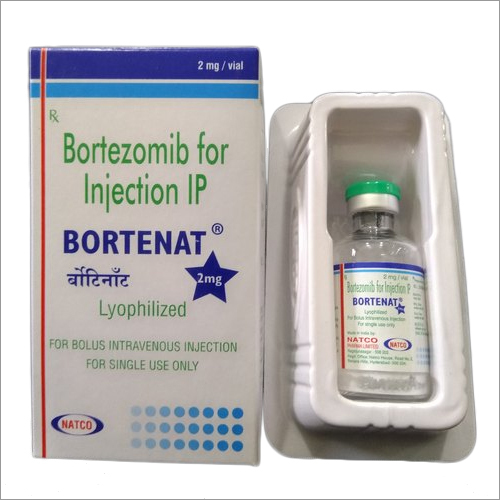

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Anti Cancer Medicine Category
Pharmaceutical Syrup
Price 100 INR / Bottle
Minimum Order Quantity : 1 Bottle
Storage Instructions : Cool & Dry Place
Dosage Form : Syrup
Enzyme Types : Other, Pharmaceutical Syrup
Origin : India
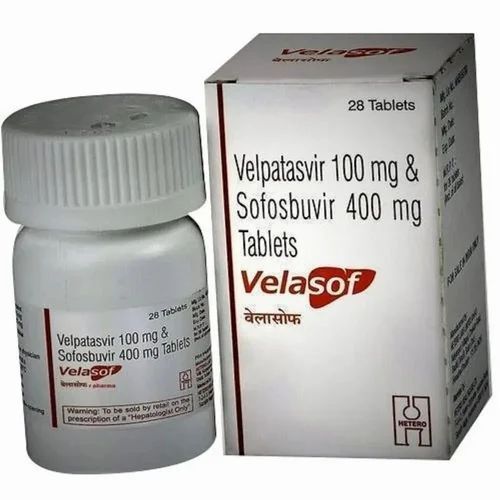
Velasof 400mg Tablets
Price 16500 INR / Pack
Minimum Order Quantity : 10 Packs
Storage Instructions : Cool & Dry Place
Dosage Form : Tablet
Enzyme Types : Other, NS5A inhibitor (Velpatasvir), NS5B polymerase inhibitor (Sofosbuvir)
Origin : India
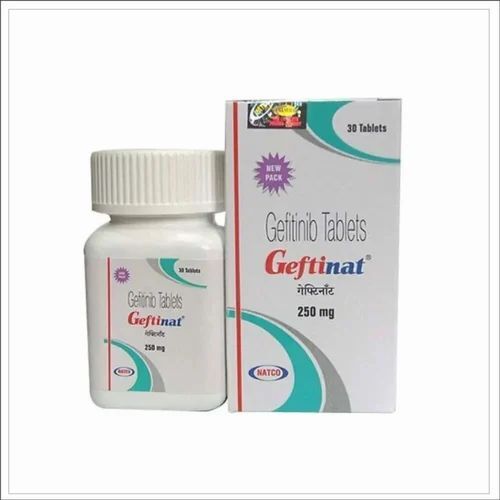
Geftinat Tablets
Price 5500 INR / Pack
Minimum Order Quantity : 100 Packs
Storage Instructions : Cool & Dry Place
Dosage Form : Geftinat Tablets
Enzyme Types : Other, Geftinat Tablets
Origin : India
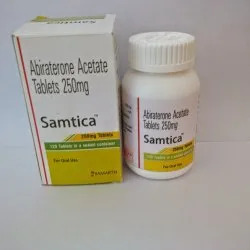
250mg Samtica Abiraterone Acetate Tablets
Price 8000 INR / Bottle
Minimum Order Quantity : 50 Bottles
Storage Instructions : Store below 30C, protect from moisture
Dosage Form : Tablet
Enzyme Types : Other, Nonenzymatic